Background
This project addresses the challenge of accurately modeling and optimizing fluid-particle interactions, which play a critical role in industries ranging from healthcare to energy and chemical processing. Dry powder inhalers (DPIs) were selected as the demonstration case due to their importance in treating asthma and Chronic Obstructive Pulmonary Disease (COPD), and because current designs suffer from inefficiencies such as high depositional losses and inconsistent performance for children and other vulnerable patients. Developing better tools to understand particle-laden flows will improve DPI performance while creating a scalable framework adaptable to other high-impact applications.
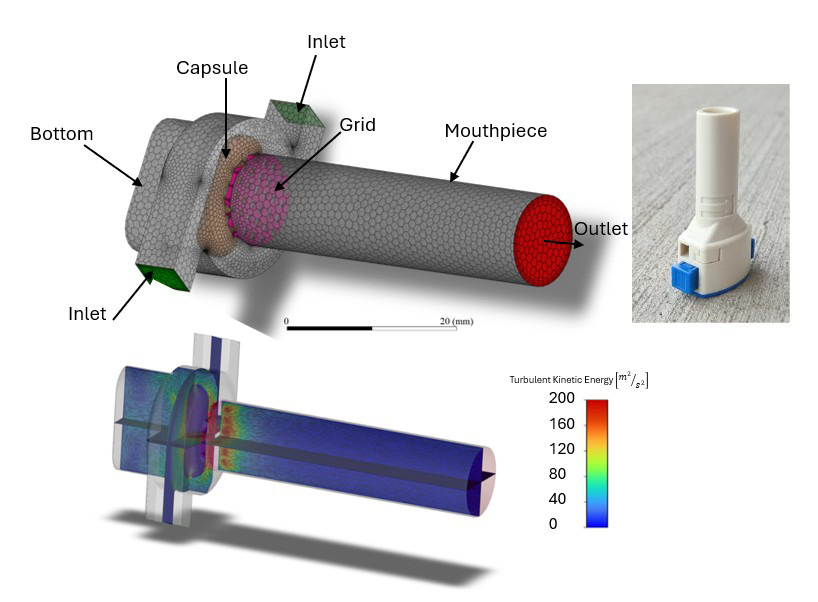
Figure 1: Geometry of the Aerolizer® dry powder inhaler model showing capsule, inlet, and mouthpiece sections with applied boundary conditions, along with preliminary CFD results of airflow distributions.
Approach
The project aims to develop and validate a coupled computational fluid dynamics-discrete element method (CFD-DEM) framework, supported by experimental testing, to simulate dense particle-laden flows with realistic particle geometries and interactions. The methodology combines literature review, solver benchmarking (ANSYS Fluent, Flow3D, MFiX-DEM), computer-aided design-driven design iterations, and in-vitro testing of 3D-printed prototypes. The framework leverages Graphics Processing Unit (GPU) and Central Processing Unit (CPU) parallelization for scalability and integrates detailed modeling of van der Waals, electrostatic, and collision forces to capture aerosolization and deposition in inhalers.
Accomplishments
During the first half of the project, the team completed an extensive literature review, developed computer-aided design models, and generated initial flow simulations through meshing and solver evaluation. Several computational solvers were tested, and MFiX-DEM was advanced as a scalable open-source option, now producing early results. In parallel, experimental setups were defined, and test rigs are being prepared to validate computational predictions. Together, these efforts have established the foundation for integrated simulation and validation cycles that will drive the next phase of inhaler design optimization.
For more information, visit Computational Fluid Particle Dynamics Solutions.
