Background
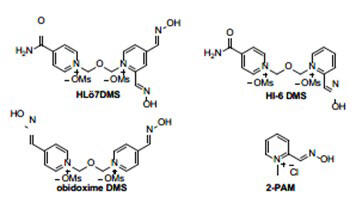
Figure 1: Lead Oxime OPNA Antidotes Neurotransmitters
The current medications used to treat organophosphorus nerve agent (OPNA) poisoning, atropine (1) and 2-PAM, provide limited protection against OPNA exposure. Other oximes, such as HLö7, HI-6, and obidoxime provide broad protection against OPNA exposure and are promising replacements (Figure1). However, their current synthesis requires BCME or BMME, which is toxic, difficult to procure, and only yields small amounts of the desired oxime antidotes. SwRI has developed a suite of key technologies to circumvent these issues that have demonstrated scalable synthesis of HLö7•2Cl for the Department of War (DoW).
The purpose of this project is to achieve ≥ 98% purity on the OMs salt form for HLö7 and HI-6, achieve ≥ 98% purity on the chloride salt form for obidoxime, demonstrate the process on a reasonable scale (~ 50 g), and develop additional technologies to ensure the future commercial feasibility of these oximes.
During our previous work on the scale-up of HLö7, we were able to achieve ~ 96% purity. In order to improve on this, we are 1) investigating the recrystallization of the Cl salt, 2) optimizing the ion pair extraction to purge impurities, and 3) optimizing the reaction yield to suppress impurity formation. Once this is accomplished, we will begin demonstrating the improved process by synthesizing HLö7, HI-6, and obidoxime on ~ 50 g scale. The ion exchange to the relevant OMs salt will be done via resin exchange initially, then a resin-free ion exchange will be developed to boost the future commercial viability of the process. Finally, to vet our process for prospective clients, we will develop an analytical method that can detect possible alkyl mesylate impurities in the oximes, which are ICH class M7 mutagenic impurities, which must be strictly controlled.
Approach
We were able to optimize our synthesis method and find the source of the impurities: polymerization during the first reaction leads to impurities in the second reaction. We optimized the ion-pair extraction to purge impurities, which now provides crude HLö7 in ~ 90% purity (up from 70%). Then we used the design of experiments to optimize the crystallization, finding that low volume formic acid had the best compromise of yield and purity. Furthermore, we modified the ion exchange to the OMs salts to provide the desired oximes with high recovery. Demonstration of the process was done on HLö7, HI-6 and obidoxime at 50 g scale. Finally, we developed a GC-MS analytical method to detect common alkyl mesylates.
Accomplishments
Over the course of the project, we were able to accomplish the following:
- Developed and patented a resin-free ion exchange
- Synthesized ~ 50 g of HI-6 at ≥ 98% purity
- Synthesized ~ 50 g of Obidoxime at ≥ 98% purity
- Synthesized ~ 50g of HLö7 at ≥ 98% purity
- Developed an analytical procedure to detect alkyl mesylates
