Background
Cryocoolers have been used for a variety of space, military, medical, semiconductor fabrication, and superconducting electronics applications. For example, cryocoolers are used to cool infrared (IR) sensors in satellites (military), superconducting magnet coils in MRI systems (medical), and astronomical detectors (space). Another application in space exploration is the cryotrapping of trace level volatile gases followed by desorption of said gases for increasing sensitivity and improving the probability of detection. The MAss Spectrometer for Planetary EXploration (MASPEX) on the Europa Clipper mission (hereafter referred to as MASPEX-Europa) is equipped with a cryocooler to cryotrap gases during a Europa flyby. After the flyby, MASPEX-Europa is sealed (valve is closed) and the cryocooler is turned off to desorb the cryotrapped gases and ionize and mass analyze the gaseous components. However, major differences in concentration (composition) of gases from the percent level to parts per billion (ppb) levels and mass spectral overlap of potential components complicate this type of measurement. This IR project aims to investigate the use of thermal desorption and/or laser desorption for selective, or stepwise, desorption of cryotrapped species liberated for mass spectral analysis, thereby improving sensitivity and limits of detection of all volatiles.
Approach
To test the capability of the cryocooler for selective, or stepwise, desorption a high surface area Mott filter (referred to as the adsorber) is attached to the cryocooler coldfinger for large trapping capacity. After the adsorber head, carbon resistors are mounted to resistively heat the adsorber head for thermal desorption experiments. The cryocooler experimental setup is incorporated into a vacuum chamber housing a mass spectrometer for mass spectral analysis of the desorbed components. A second experimental setup was employed without the Mott filter to cryotrap on the cryocooler cold finger surface to: 1) simplify the setup; 2) reduce the thermal mass load to achieve lower cryotrapping temperatures. A close-up photograph of the components mounted on the cryocooler are displayed in Figure 1. The mass spectrometer employed for these experiments is a time-of-flight (TOF) mass spectrometer referred to as the W-TOF-MS for the W-shaped flight path (trajectory) that the ions take in a folded ion optics, dual reflectron design. This W-TOF-MS is being developed under NASA’s Heliophysics Technology and Instrument Development for Science (HTIDS) program and is used in this IR effort to demonstrate the applicability of the cryocooler to future instruments and potential space missions beyond MASPEX-Europa. The TOF MS is essential with its fast repetition rate and collection of the entire mass spectrum with each extraction (Fellgett Advantage) to enable the detection of the rapidly changing desorbed gas composition.
Laser thermal desorption from the cryocooler cold finger surface was attempted for some of the cryotrapped species. Some evidence of laser thermal desorption was observed but reproducible and reliable results were not achieved. Higher laser power (increased fluence, or energy per area) would aid this experiment.

Figure 1. Photograph of the components mounted on the cryocooler cold finger for the thermal desorption experiments using the first experimental setup (left). Photograph of the simplified, second experimental setup (right).
Accomplishments
Several accomplishments were achieved over the course of the project:
- Incorporation of all components on the cryocooler (adsorber, temperature sensors, heating resistors)
- Temperature calibration of the temperature sensors
- Data obtained for cryotrapping and desorption of a variety of volatiles including atmospheric gases, organics, hydrocarbons (alkanes), and aromatic species.
- Resolution values (as a function of temperature) determined from alkane thermal desorption experiments
- Demonstrated proof-of-performance of stepwise thermal desorption of cryotrapped volatiles for improved sensitivity and detectability that can be implemented on MASPEX-Europa and future space missions
- Demonstrated linearity of response from experiments with varying concentrations of pentane
A summary of cryotrapping and desorption data for a variety of volatiles are displayed in Figure 2. The thermal desorption experiment shows resolution of the alkanes and that step-wise desorption of components is possible from controlled heating of the adsorber. Further investigation of the selected ion current of targeted m/z ions representative of individual alkanes from the mass spectral data provided by the W-TOF-MS confirm the stepwise desorption and resolution of a cryotrapped alkane mixture (Figure 3). Linearity of response has been established from varying concentrations (pressure) of pentane being cryotrapped and subsequently desorbed from the adsorber with the data presented in Figure 4.
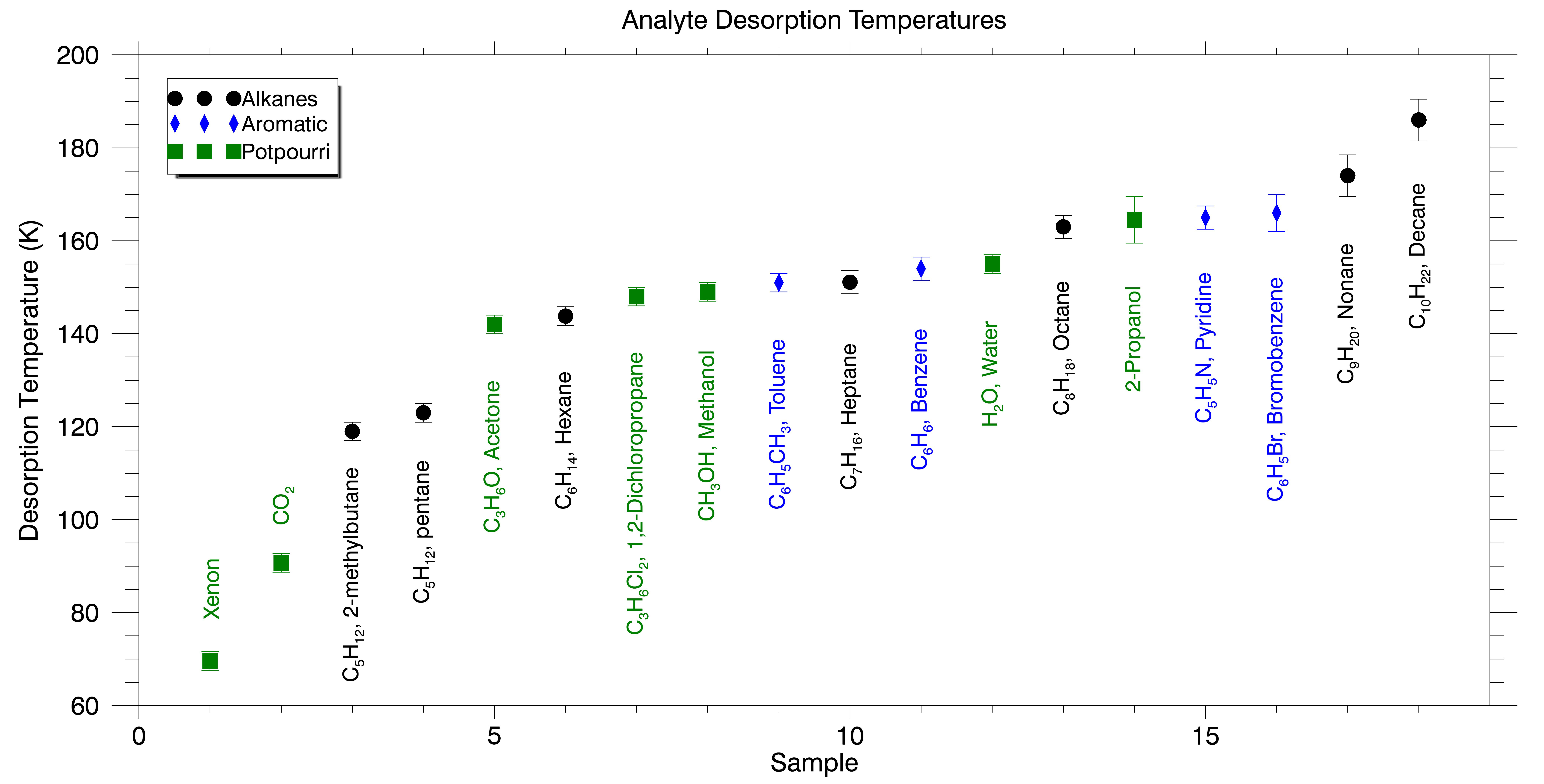
Figure 2. Summary desorption temperature data for the analytes tested during this project.
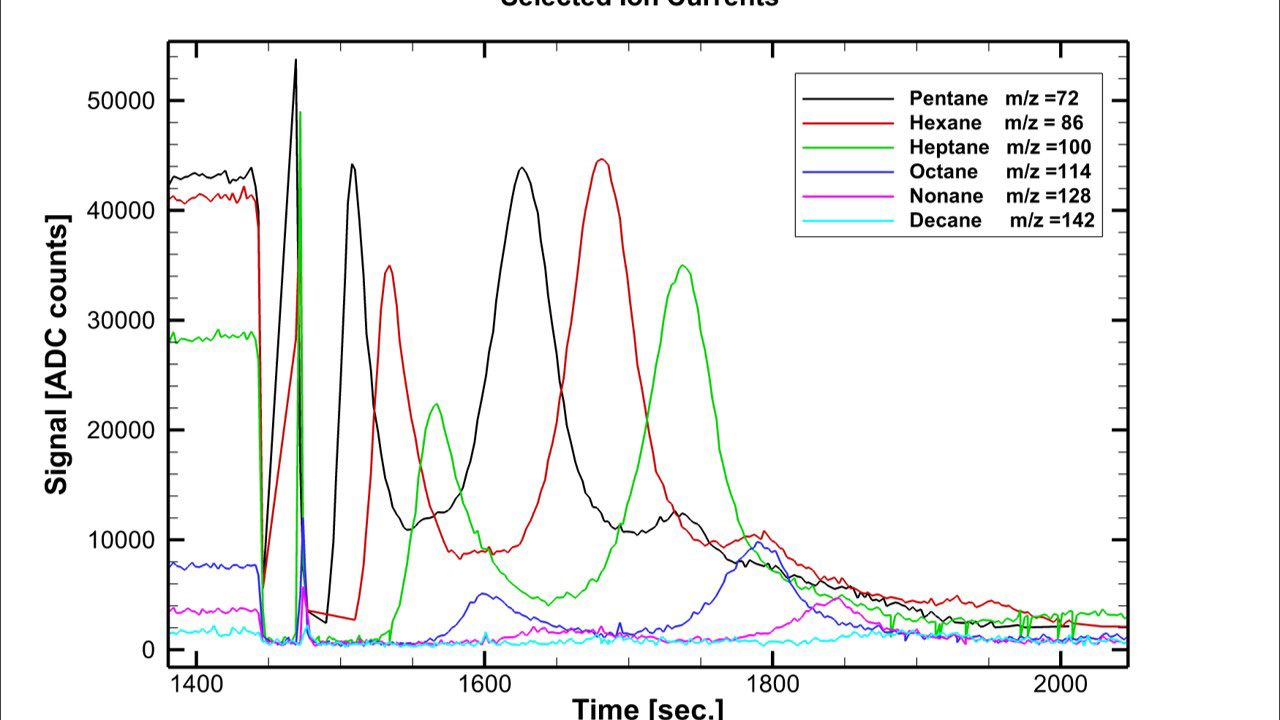
Figure 3. Selected ion currents, or targeted m/z values, representing pentane (m/z = 72, black), hexane (m/z = 86, red), heptane (m/z = 100, green), octane (m/z = 114, blue), nonane (m/z = 128, magenta), and decane (m/z = 142, cyan) tracked with W-TOF-MS display the step-wise desorption (resolution) of the individual alkanes from a cryotrapped alkane mixture.
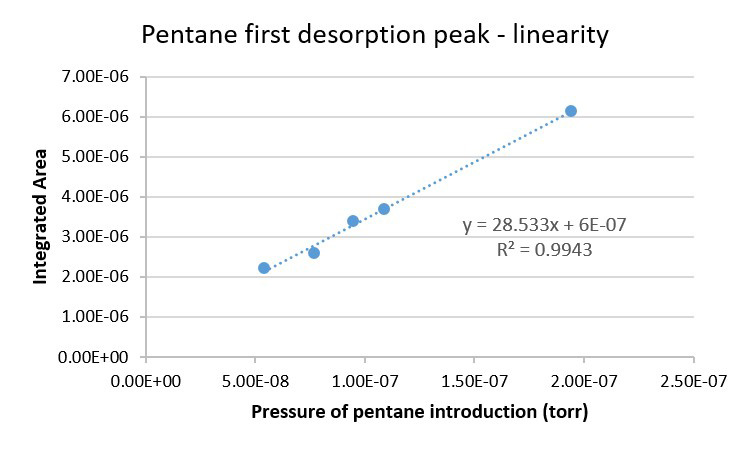
Figure 4. Linearity of response data for pentane obtained by integrating the area of the desorption curves at different concentrations (pressure).
